Hi all,
After building several successful lead/alum cells from old 12V gel cell batteries and testing my alum/water mix... I decided to try to make my first alum battery using a new Napa GS 12 volt (8 Ah) motorcycle battery.
I mixed the alum with warm distilled water in a blender to a nice constancy, "just a bit slippery between the fingers" , so I think it is good. I let the battery sit for about an hour before I applied the first charge, to ensure that the cells were evenly saturated. Looking back....I decided to charge the battery up on my SG energizer for its first charge, so I think the plates may not have been formed correctly.
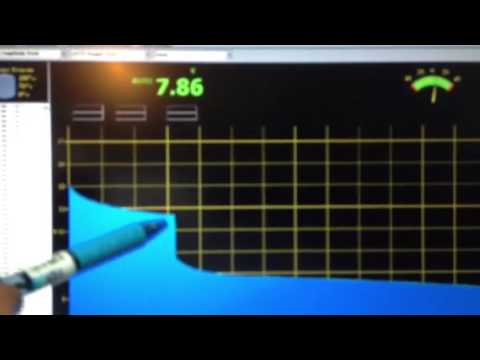
I dont seem to be getting the final open circuit voltage to hold above 10.75 VDC. The battery takes a charge to about 13.99 volts, then drops off to 10.75 volts when the charger is removed. I tested the capacity with at load at C10 (1.2 amps) and the voltage just drops off, dramatically within 30 minutes.
Any help would be appreciated, thank you
After building several successful lead/alum cells from old 12V gel cell batteries and testing my alum/water mix... I decided to try to make my first alum battery using a new Napa GS 12 volt (8 Ah) motorcycle battery.
I mixed the alum with warm distilled water in a blender to a nice constancy, "just a bit slippery between the fingers" , so I think it is good. I let the battery sit for about an hour before I applied the first charge, to ensure that the cells were evenly saturated. Looking back....I decided to charge the battery up on my SG energizer for its first charge, so I think the plates may not have been formed correctly.
I dont seem to be getting the final open circuit voltage to hold above 10.75 VDC. The battery takes a charge to about 13.99 volts, then drops off to 10.75 volts when the charger is removed. I tested the capacity with at load at C10 (1.2 amps) and the voltage just drops off, dramatically within 30 minutes.
Any help would be appreciated, thank you


 as a Lead-Acid Battery....
as a Lead-Acid Battery....
Comment